|
Methods: KEYNOTE-048 was a randomised, phase 3 study of participants with untreated locally incurable recurrent or metastatic HNSCC done at 200 sites in 37 countries. 
Go to our website and learn all the details of the KEYNOTE-048 study, presented at the European Society for Medical Oncology (ESMO) Virtual Congress in September. Press Briefing 4, October 22 - Head & Neck CancerLBA8PR - KEYNOTE-048: Phase 3 study of first-line pembrolizumab (P) for recurrent/metastatic head and neck. Background: Pembrolizumab is active in head and neck squamous cell carcinoma (HNSCC), with programmed cell death ligand 1 (PD-L1) expression associated with improved response. The publication of the first results of the KEYNOTE-048 study in 2019 has transformed the standard treatment for metastatic and recurrent disease, according to Fernanda Bohns Pruski Ramos, an oncologist at OC Oncology Center HSL PUCRS, a clinic of the Oncoclínicas Group in Rio Grande do Sul: “This changed the treatment because we observed a benefit in overall survival when we added immunotherapy (in this case, pembrolizumab) to platinum and fluorouracil,” she says. 
The data come from the KEYNOTE-048 study.Īccording to Daniel Oliveira Brito, oncologist at the Bahia Ondina Oncology Center, a clinic of the Oncoclínicas Group in Salvador, the treatment of metastatic head and neck squamous cell carcinoma had been going on for about 12 years without news for the first-line scenario: “The combination of platinum chemotherapy (platinum doublet) with fluorouracil plus cetuximab (evaluated by the EXTREME clinical trial) was the standard, showing median overall survival at 10.1 months and response rate of 36%,” Brito said. Long-term follow-up of patients with recurrent or metastatic head and neck squamous cell carcinoma has confirmed the advantage of pembrolizumab, with or without chemotherapy, over the regimen consisting of cetuximab and chemotherapy for PD-L1-positive patients. The randomised, open-label, phase 3 KEYNOTE-048 study of pembrolizumab given alone or in combination with a chemotherapy regimen of platinum and 5-fluorouracil establishes anti-programmed cell death 1 (PD-1)-based therapy as a first-line treatment option for patients with locally incurable recurrent or metastatic HNSCC. What this drug does is block PD-1 and thus make the cancer cells visible again to the immune system, so that it can attack them freely. This linkage prevents the T-cell from attacking the PD-L1 carrying cell, so that certain cancer cells become “invisible” to attacks by the immune system. 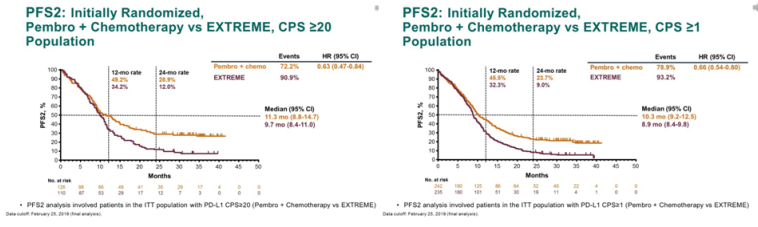
PD-1 binds to another protein, PD-L1, that exists on certain cells in the body, including some of the cancerous types. Further clinical interpretation and implications of KEYNOTE-048 findings. The P-III KEYNOTE-048 study results: CPS20 & CPS 1 for combination- 40 & 35 reduction in death OS (14.7 & 13.6 vs 11 & 10.4 mos.) ORR (42.9 & 36.4. Pembrolizumab is an immunotherapy that targets a protein called PD-1, found on T cells in the immune system. Keynote for Mac, iOS and iCloud lets you make dazzling presentations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
